UV Light Selection: What is the difference between UV-A,UV-B and UV-C?
Can a wound be disinfected with a blacklight? An expert might find the question ridiculous, yet it reveals a prevalent and harmful misunderstanding. We utilise UV light to cure coatings, find leaks, and even tan skin because we know it can kill microorganisms. How is it possible for one kind of light to accomplish all of these tasks? It can't, is the response. Believing that "UV is UV" is a mistake that can, at best, result in a failed application and, at worst, a major safety hazard.
The wavelength is the crucial distinction. Energy, penetrating power, and the particular biological or chemical effect are all determined by wavelength. This manual will give you a clear, scientifically grounded foundation for understanding UVA, UVB, and UVC-not just their distinctions, but also how to use that understanding to choose and employ the best equipment for your work.
What we will discuss is as follows:
The basic principle: each UV type is defined by its wavelength.
practical B2B applications that go beyond the fundamentals.
a framework for safety and risk assessment that cannot be compromised.
A realistic approach to choosing a UV system.
Let's begin with that basic principle.
1. The Essential Distinction: Wavelength Is Everything
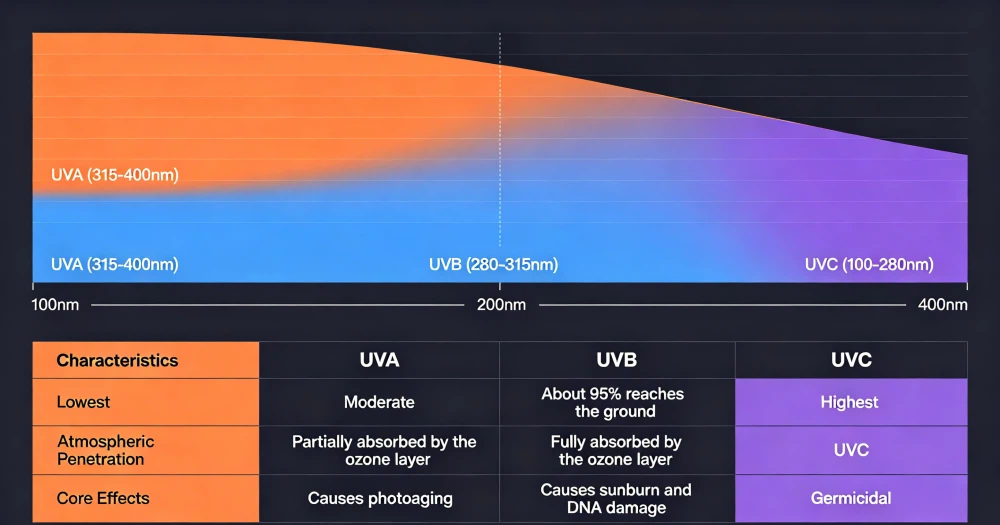
The wavelength of UV light, expressed in nanometres (nm), is the first and last characteristic to comprehend. Everything else is determined by this one value. The following spectral boundaries serve as a foundation for the discussion:
UVA: 315–400 nm
UVB: 280–315 nm
UVC: 100–280 nm
For comparison, the thickness of a human hair is approximately 75,000 nanometres. The results of these particular slices of the invisible spectrum vary greatly. Higher energy is found in shorter wavelengths. This directly relates to their fundamental traits.
| Characteristic | UVA (315-400nm) | UVB (280-315nm) | UVC (100-280nm) |
|---|---|---|---|
| Relative Energy | Lowest energy | Medium energy | Highest energy |
| Atmospheric Penetration | Nearly 95% reaches the Earth's surface. | Partially absorbed by the ozone layer. | Completely absorbed by the ozone layer; does not reach the surface naturally. |
| Primary Biological/Physical Effect | Penetrates deeply into the dermis, causing aging; initiates photochemical reactions for curing and fluorescence. | Damages the epidermis, causing sunburn and directly damaging DNA. | High energy disrupts DNA/RNA of microorganisms, rendering them unable to replicate (germicidal effect). |
To put it briefly, energy determines capacity, and wavelength determines energy. This key opens the door to comprehending why each type of UV performs exceptionally well in entirely different contexts.
2. Not All UV Is Used for Disinfection: Essential Uses Described
The usefulness of UV light in the real world is entirely dependent on matching the right wavelength to a particular task.
The Pathogen Inactivator: UV-C
The main professional purpose of UV-C is disinfection. Its physical process involves the absorption of UVC photons at the peak wavelength of 265 nm by the DNA and RNA of bacteria, fungi, and viruses. By fusing genetic base pairs (a process known as dimerisation), this energy taints the pathogen's blueprint, preventing it from reproducing or spreading infection. It is a focused energy hit rather than a chemical toxin.
This idea has important applications in a variety of industries:
Healthcare: N95 respirator sterilisation, terminal room disinfection, and airborne pathogen prevention in operating rooms.
HVAC & IAQ: To save energy costs and enhance air quality, business facilities should clean their air handler coils and disinfect their moving airstreams.
Water treatment: Chemical-free disinfection for household point-of-use systems, pharmaceutical-grade process water, and municipal water.
Life Sciences: Surface sterilisation for cell culture research in CO2 incubators.
An important distinction is that an ozone-generating lamp (185 nm) is not the same as UV-C (254 nm from a low-pressure mercury lamp or LED). Pathogens are rendered inactive by UV-C; ozone offers an extra chemical deodorising benefit but necessitates stringent ventilation procedures. Don't mix up the two.
UV-A: The Workhorse of Industry
UV-A is the silent workhorse in industrial and scientific contexts, while UV-C dominates the discourse on disinfection. Although it effectively initiates photochemical reactions, its lower energy does not damage DNA.
There are two particularly noteworthy applications:
Fluorescence and NDT: Some materials fluoresce in the visible spectrum when exposed to UV-A. This is essential for forensic investigation, HVAC leak detection using fluorescent dyes, and non-destructive testing (NDT) to find engine component defects. In these situations, 365 nm is frequently chosen over 395 nm due to its "cleaner," less noticeable purple tint.
UV Curing: In carefully designed adhesives, coatings, and inks, UV-A starts the polymerisation process. UV-A curing is used in industries including automated optical bonding, 3D printing, and electronic component encapsulating because of its quickness, accuracy, and minimal heat impact.
The Speciality Niche: UV-B
A smaller yet crucial function is played by UVB, mainly in the life sciences and health. It is the only wavelength that can start the synthesis of vitamin D. Narrowband UVB is an essential phototherapy technique for treating diseases like vitiligo and psoriasis in a controlled medical setting. It is used as a controlled environmental stressor in plant research to investigate the resilience of plants.
Knowing how powerful each form of UV is is only half the story. Since using tremendous power without strict safety precautions is a liability, we now have to face the risks head-on.
3. The New Frontier of Far-UVC, Risk, and Safety
A methodical strategy to controlling biological dangers and a sober awareness of them are necessary for professional UV use.
The Risks according to Wavelength
The harm is not intangible. UVC's strong energy causes rapid skin erythema and photokeratitis, a painful burn to the cornea that feels like sand in the eyes. Unprotected eyes are particularly sensitive, but it mostly affects the outermost, dead layer of skin. Sunburn and the majority of skin malignancies are caused by UVB, which is a direct DNA sniper. By penetrating deeper into the dermis, UVA acts as a slow-moving ageing agent and indirectly damages DNA, increasing the risk of long-term cancer. Although all UV light is thought to be carcinogenic, each has a different mechanism and tissue target.
UV-C Safety: A Checklist of Golden Rules
Invisible light cannot be detected by instinct. The following should be required by a documented Standard Operating Procedure for any commercial or industrial UV-C deployment:
People: Full-coverage clothing and dedicated polycarbonate face shields are essential. Wrists must be covered by gloves. It is necessary to control and limit access to the region.
Equipment: Safety interlocks are required for all high-intensity systems. To confirm dose, use a calibrated UV radiometer rather than speculating. If necessary, install viewing windows made of acrylic or another UV-blocking material.
Environment: When operating, make sure the space is empty. Ventilate properly to remove any ozone and exhaust heat from non-LED sources. Post a detailed incident response procedure.
222nm Far-UVC: A Safer Future
With the advent of filtered 222nm Far-UVC technology, the safety equation is evolving. The idea is straightforward but profound: 222 nm light is so heavily absorbed by organic material that it cannot reach living cells through the tear layer on the surface of the eye or the outer layer of dead skin cells. These layers can be penetrated by conventional UVC (254 nm). This enables disinfection in occupied spaces with a significantly improved safety profile, according to early scientific consensus. Cost, power density, and bulb life are still changing, so it's not a panacea, but it's a real safety revolution to observe.
We have given you the understanding of safety, applications, and science. Let's now transform this into a useful, repeatable method for making decisions.
4. Your Choice Process: From Requirement to Definition
Avoid using a product catalogue when choosing a UV system. Review your own needs in a methodical manner to get started.
Step 1: Identify Your Main Objective
Clearly state what your main goal is. Is it disinfection (eradicating pathogens on a surface, in water, or in the air)? Or is it curing (polymerising an ink or adhesive)? A particular UV band will be clearly indicated by a particular application.
Step 2: Convert the Objective into Crucial Details
Understanding UV Dose, expressed in millijoules per square centimetre (mJ/cm2), is necessary for disinfection applications. Dose = Intensity x Time is a straightforward formula. To achieve a 4-log (99.99%) reduction in a target organism such as E. coli, a certain dose is needed. You must balance lamp intensity with conveyor speed or water flow rate in order to achieve that dosage. Peak Irradiance (W/cm²) and making sure the lamp's peak wavelength exactly matches your photoinitiator's reactivity are crucial parameters for a curing application.
Step 3: Contrast LED and Lamp Product Forms
The light source itself is frequently the ultimate practical decision. UV LEDs have a long, stable lifespan with narrow-band output that is energy-efficient for particular wavelengths, instant on/off switching, and a mercury-free footprint. For certain very high-power, wide-area applications where initial capital cost is the only consideration, a conventional mercury lamp is still a viable option. LED is the best, contemporary instrument for the majority of industrial and disinfecting operations that are precise, controlled, and regularly switched.
5. Use Light Respectfully
The distinction between UVA, UVB, and UVC is the scientific basis for safe, efficient application; it is not an academic exercise. The application is your objective; safety is your non-negotiable boundary. UV light is a very potent physical tool, but its real worth comes from the skill and diligence with which you use it.
Are you prepared to use this framework for your particular problem? For a free, no-obligation project consultation, speak with one of our UV application experts.
FAQ
Q: Q: Can I use a UVA lamp for disinfection?
A: A: No. UVA has insufficient energy to cause the critical DNA damage required to inactivate pathogens. Only UVC, and specifically at the germicidal wavelength near 265nm, is effective for this purpose.
Q: Q: How do I safely use a UV-C disinfection lamp in a professional setting?
A: A: Never expose unprotected skin or eyes. Always wear polycarbonate face shields and protective clothing. Use physical interlocks and warning signs, and ensure the space is unoccupied during operation. Always follow a documented safety protocol.
Q: Q: Which is better, a UV LED or a traditional mercury lamp?
A: A: For most modern applications, UV LEDs are superior. They offer instant cycling, contain no mercury, have a longer lifetime, and their narrow-band output is more energy-efficient. Mercury lamps may still be found in older, high-power broad-spectrum installations.
Q: Q: Does UVC light produce ozone?
A: A: Standard germicidal UVC at 254nm does not produce ozone. Ozone is generated by shorter wavelengths, specifically 185nm, emitted by some specialty quartz lamps. If your system uses 185nm lamps, enhanced ventilation is mandatory. This is a critical specification to verify.
Contact
Kevin Rao
Email:bwzm12@benweilighting.com
Tel/Whatsapp:+8619972563753









