The Four Components of a Li-ion Battery
Many household appliance juggernauts are currently scrambling to release cordless stick vacuum cleaners.
Many people adore this appliance since it is light enough for even a youngster to use while still having excellent suction strength.
Li-ion batteries made it feasible for cordless vacuums to be created, in major part.
High energy density yet being light Li-ion batteries are more efficient and have a higher capacity than conventional batteries.
are frequently used in a variety of fields, including power tools, energy storage systems, tiny appliances, and IT equipment.
as well as electric cars.
Today, let's examine Li-ion batteries from top to bottom.
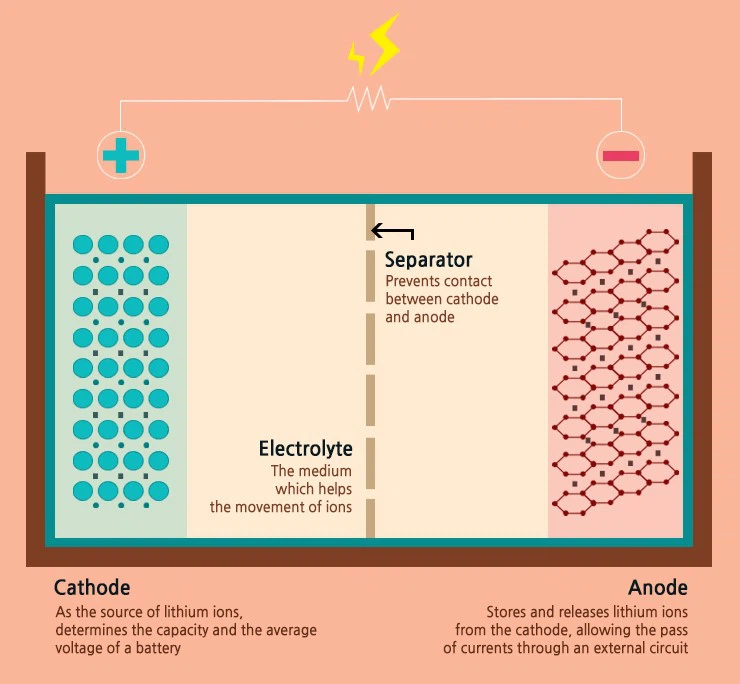
Cathode, Anode, Electrolyte, and the Li-ion battery consist of four components.
Separator
Cathode, anode, electrolyte, and separator are the four essential components of a lithium-ion battery.
A Li-ion battery needs each and every component since it cannot operate if one of them is absent.
A Li-ion battery's "cathode" influences its capacity and voltage.
The lithium in a lithium-ion battery undergoes chemical processes to produce power.
For this reason, lithium is naturally introduced into the battery, and the area where the lithium is located is known as the "cathode."
Lithium oxide is utilised for cathodes because lithium in its elemental form, which consists of lithium and oxygen, is unstable.
The term "active material" refers to a substance like lithium oxide that blocks the electrode reaction of the real battery.
In other words, lithium oxide serves as the active component in the cathode of a Li-ion battery.
A thin aluminium foil used to support the cathode's coated frame may be seen if you look at the cathode closely.
using a mixture of an active substance, a conductive additive, and a binder.
Lithium ions are present in the active substance, and a conductive additive is added to improve conductivity;
Additionally, the binder serves as an adhesive to aid in the proper adhesion of the conductive additive and active material to the aluminium substrate.

The battery's properties are heavily influenced by the cathode.
because the cathode's active material type affects the battery's voltage and capacity.
The capacity increases with the quantity of lithium present, and the voltage rises with the magnitude of the potential difference between the cathode and the anode.
Depending on the kind, the potential difference between anodes and cathodes is typically minor for anodes and somewhat significant for cathodes.
As a result, the cathode is crucial in determining the battery's voltage.
"Anode" transmits electrons down a wire.
The anode substrate is covered with active material, just like the cathode.
The function of the anode's active substance is to permit electric current to pass via the external circuit.
Lithium ions emitted from the cathode can be absorbed or released reversibly while doing so.
Lithium ions are kept in the anode, not the cathode, when the battery is being charged.
When the cathode and anode are now connected by a conducting wire (in a discharge condition),
The electrolyte naturally allows lithium ions to return to the cathode,
and the lithium ions' separated electrons (e-) flow down the wire while producing power.
Use of stable-structured graphite for anodes and coating of active material on the anode substrate
a binder and a conductive additive.
Graphite's ideal characteristics, namely its structural stability and low electrochemical reactivity,
The material is thought to be appropriate for use as an anode given its affordability and capacity to store large amounts of lithium ions.
"Electrolyte" solely permits ion mobility.
It was said that lithium ions flow through the electrolyte while discussing cathode and anode.
and the wire is filled with electrons.
This is essential for allowing a battery to consume power.
We won't be able to use electricity and our safety will be in danger if ions pass through the electrolyte.
The element that fulfils this crucial function is the electrolyte.
It acts as the conduit that allows only lithium ions to travel back and forth between the cathode and the anode.
High ionic conductivity materials are primarily utilised for the electrolyte so that lithium ions may travel back and forth readily.
There are salts, solvents, and additives in the electrolyte.
Lithium ions flow through the salts, which are dissolved in organic liquids called solvents.
and for certain objectives, the additives are introduced in limited quantities.
This method of making electrolyte prevents electrons from passing through and only permits ions to flow to the electrodes.
Additionally, the kind of electrolyte affects how quickly lithium ions migrate.
Therefore, only electrolytes that adhere to strict requirements can be utilised.
"Separator," the impermeable partition separating cathode and anode
The electrolyte and separator define a battery's safety, while the cathode and anode determine a battery's fundamental performance.
Separator keeps cathode and anode apart by acting as a physical barrier.
It carefully allows just the ions to pass through the interior tiny hole while obstructing the direct passage of electrons.
It must thus meet all of the physical and electrochemical requirements.
Today's synthetic resin separators, such polyethylene (PE) and polypropylene (PP), are commercially available.
We have examined the four key factors that affect how well Li-ion batteries function thus far.
Currently, Samsung SDI is stepping up research and development of novel materials for improving battery performance.
while steadfastly carrying on its efforts to enhance the functionality of current materials and key technologies.
Through the development of high-capacity, high-efficiency Li-ion batteries,
Samsung SDI wants to lead the way in the development of batteries that will improve the quality of life for people all over the world.




